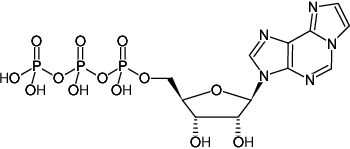
(1,N6-Etheno-ATP)
1,N6-Etheno-adenosine-5'-triphosphate, Sodium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-1103S | 500 μl (10 mM) | 72,30 | Add to Basket/Quote Add to Notepad |
| NU-1103L | 5 x 500 μl (10 mM) | 209,70 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C12H16N5O13P3 (free acid)
Molecular Weight: 531.20 g/mol (free acid)
Exact Mass: 531.00 g/mol (free acid)
CAS#: 60777-99-3
Purity: ≥ 95 % (HPLC)
Form: solution in water
Color: colorless to slightly yellow
Concentration: 10 mM - 11 mM
pH: 7.5 ±0.5
Spectroscopic Properties: λmax 275 nm, ε 6.0 L mmol-1 cm-1 (Tris-HCl pH 7.5), λexc 300 nm, λem 415 nm
Applications:
Fluorescence quenching[1]
Exchange rate from G-actin and wild type[2]
FRET-studies on conformational changes[3]
Formation in erythrocytes andendothelial cells[4]
Fluorescence anisotropy changes during protein binding[5]
Influence on centromeric binding protein E associated activity[6]
Specific Ligands:
Yeast and muscle actins[7]
Coflin and profilin[8]
P2Y receptor in cardiac endothelial cells[9]
BIOZ Product Citations:
Selected References:
[1] Saratovskikh et al. (2010) Kinetic of transport of technogenic oxicants through model membranes. J. Environmental Protection 1:410.
[2] McKane et al. (2006) Effect of the substitution of muscle actin-specific subdomain 1 and 2 residues in yeast actin and actin function. J. Biol. Chem. 281:29916.
[3] Dedova et al. (2006) Thymosin β4 induces a conformational change in actin monomers. Biophysical J. 90:985.
[4] Mattig et al. (2003) Modulation of adenine nucleotide concentrations in human plasma by erythrocytes and endothelial cells. Thrombosis Res. 110:195.
[5] Suarez et al. (202) Biochemical defects in retina-specific human ATP binding cassette transporter nucleotide binding domain 1 mutants associated with macular degeneration. J. Biol. Chem. 277:21759.
[6] Thrower et al. (1995) Mitotic HeLa cells contain a CENP-E associated minus end-directed microtubule motor. EMBO Journal 14:918.
[7] Wen et al. (2008) Control of the ability of profilin to bind and facilitate nucleotide exchange from G-actin. J. Biol. Chem. 283:9444.
[8] Kardos et al. (2009) The effects of ADF/cofilin and profilin on the conformation of the ATP-binding cleft of monomeric actin. Biophysical J. 96:2335.
[9] Yang et al. (1994) Purinergic axis in cardiac blood vessels: Agonist mediated release of ATP from cardiac endothelial cells. Circ. Res. 74 (3):401.
Singh et al. (2011) Crystal structures explain functional differences in the two actin depolymerization factors of the malaria parasite. J. Biol. Chem. 286 (32): 28256.
Aguilar et al. (2001) Ectoenzymatic breakdown of diadenosine polyphosphates by Xenopus laevis oocytes. Eur. J. Biochem. 268 (5):1289.
Churchich et al. (2000) A catalytic site of protein disulfide isomerase probed with adenosine-5 '-triphosphate analogs. BBA-Protein Struct. M. 1479 (1):293.
Gualix et al. (1999) Studies of chromaffin granule functioning by flow cytometry: Transport of fluorescent epsilon-ATP and granular size increase induced by ATP. Receptor Channel 6 (6):449.
Franksskiba et al. (1994) Quenching of fluorescent nucleotides bound to myosin - a probe of the active-site conformation. Biochemistry-US 33 (42):12720.
Miki et al. (1994) Domain motion in actin observed by fluorescence resonance energy-transfer. Biochemistry-US 33 (33):10171.
Root et al. (1992) The accessibility of ethenonucleotides to collisional quenchers and the nucleotide cleft in G-Actin and F-Actin. Protein Sci. 1 (8):1014.
Conner et al. (1989) Sister chromatid exchange induced by etheno-ATP derivatives invitro. Cancer Res. 49 (14):3839.
Wang et al. (1981) Exchange of 1, N (6)-etheno-ATP with Actin-bound nucleotides as a tool for studying the steady-state exchange of subunits in F-Actin solutions. P. Natl. Acad. Sci.-Biol. 78 (9):5503.
Yanagida (1981) Angles of nucleotides bound to cross-bridges in glycerinated muscle-fiber at various concentrations of epsilon-ATP, epsilon-ADP and epsilon-AMPPNP detected by polarized fluorescence. J. Mol. Biol. 146 (4):539.
Burtnick et al. (1979) Circular-polarization of the fluorescence of Actin-bound epsilon-ATP - effects of binding DNAse-I. FEBS Lett. 97 (1):166.
Kaplan et al. (1976) Mitochondrial ATPase activity and adenine-nucleotide transport with epsilon-ATP. J. Cell Biol. 70 (2):a414.
Hohne et al. (1975) New principle for activity measurement of ADP or ATP dependent enzymes - fluorescence quenching of epsilon-ADP and epsilon-ATP by divalent metal-ions. Anal. Biochem. 69 (2):607.