The Thermofluor Screens JBScreen FUNDAMENT and JBScreen SPECIFIC allow identification of protein-stabilizing buffer conditions which is pivotal for protein purification, characterization and crystallization.
Undesired overlay of screening of interdependent variables is prevented by strictly categorizing stability screening of proteins into
1. FUNDAMENTAL factors that influence the whole protein molecule (pH and ionic strength)
2. SPECIFIC factors that affect energetically important hot spots on the protein (substrates and their analogs, cations, anions, ...)
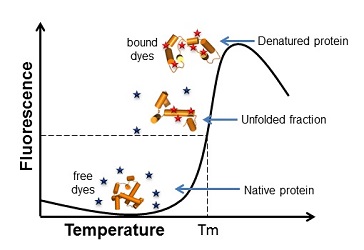
The protein’s melting temperature (Tm) is used as reporter for protein stability and determined by a thermal shift assay (96-well plate format) monitoring the unfolding of a protein in a temperature-dependent manner. The higher the Tm, the higher is the thermostability of the protein in that specific environment.
α-Chymotrypsinogen A was applied in JBScreen Thermofluor FUNDAMENT and SPECIFIC.
Stabilizing conditions (high ionic strength, neutral pH, CaCl2 & FeCl3) were combined without further optimization. A crystallization screen was set up:

Crystal growth was promoted by the stabilizing specific ions CaCl2 & FeCl3 from JBScreen Thermofluor SPECIFIC.
Talk held at HEC19 in Warberg, September 29th, 2016