For more than 2 decades we work according to the principle that modern molecular diagnostics, whether applied in scientific research or in clinical practice, can only be advanced with truly high-quality products. This quality claim is not only directed at the quality of our products, but also at our ecological responsibility and the relationships we maintain with our clients – worldwide, long-term and trusting.
We are specialized in pre-GMP production of ultrapure dNTP and NTP for PCR and alike at scales of 5,000 litres @100 mM p.a.. In addition, we manufacture customized raw materials (nucleotides, probes and enzymes up to master mixes) for (RT)PCR-based in-vitro diagnostic kits and pre-clinical API stages.
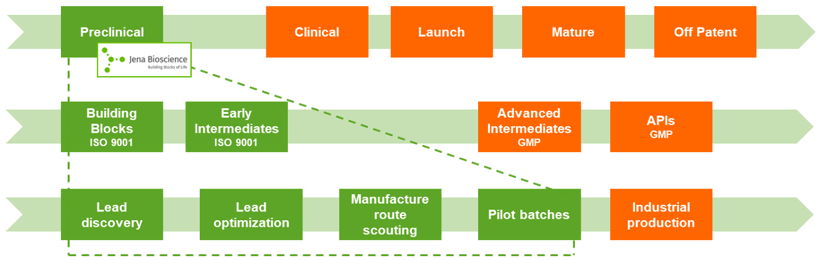
Jena Bioscience's pre-GMP manufacturing capabilities at one glance
Our long-term established and reliable processes are subject to a continuous improvement cycle and still leave room for individual requirements. By closely interlinking R&D and production in one facility in Germany, we are able to develop products in close cooperation with our clients according to customer-specific conditions. We enable customized formulations, filling and labeling (OEM).

Jena Bioscience has established a quality management system according to DIN EN ISO 9001, an environmental management system according to DIN EN ISO 14001.
We have set the course for ISO 13485 certification and already implemented relevant requirements of this ISO standards. This has been greatly acknowledged by our clients. Since 2018, we have passed all ISO 13485 customer audits without deviation.
We commit ourselves to work according to international standards and continuously improve our quality and environmental management system and our in house processes to meet our high quality standards.
Our structures and programs are designed to integrate environmental considerations throughout the company’s operations. We ensure the effective utilization of products needed for the manufacturing process and all other operations and preferably use materials that can be reused, recycled or disposed environmentally friendly. We also monitor the effective implementation of our environmental requirements and take measures accordingly. Jena Bioscience goes beyond minimum legal compliance and continuously improves its environmental performance.
Get your own impressions on site. Contact us with your questions or schedule an audit appointment.