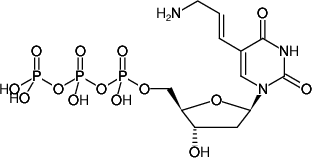
5-(3-Aminoallyl)-2'-deoxyuridine-5'-triphosphate, Sodium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-803-1 | 1 mg | 118,40 | Add to Basket/Quote Add to Notepad |
| NU-803-5 | 5 mg | 515,60 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C12H20N3O14P3 (free acid)
Molecular Weight: 523.22 g/mol (free acid)
Exact Mass: 523.02 g/mol (free acid)
CAS#: 116840-18-7
Purity: ≥ 95 % (HPLC)
Form: solid
Color: white to off-white
Spectroscopic Properties: λmax 289 nm, ε 7.1 L mmol-1 cm-1 (Tris-HCl pH 7.5)
Applications:
Incorporation into DNA/cDNA by
Description:
Aminoallyl-dUTP is recommended for two-step labeling of DNA/cDNA e.g. by PCR, Nick Translation, Primer Extension, 3'-End Labeling and Reverse Transcription. It is enzymatically incorporated into DNA/cDNA as substitute for its natural counterpart dTTP. The resulting Amine-functionalized DNA/cDNA can subsequently be labeled via the classic Amine/NHS Ester reaction that offers the choice
BIOZ Product Citations:
Selected References:
[1] Dirsch et al. (2007) Probe production for in situ hybridization by PCR and subsequent covalent labeling with fluorescent dyes. Appl. Immunohistochem. Mol. Morphol. 3:332.
[2] Cox et al. (2004) Fluorescent DNA hybridization probe preparation using amine modification and reactive dye coupling. BioTechniques 36:114.
[3] Cherkasov et al. (2010) New Nucleotide Analogues with Enhanced Signal Properties. Bioconjugate Chem. 21 (1):122.
[4] Unciti-Broceta et al. (2003) The use of solid supports to generate nucleic acid carriers.Accounts of Chemical Research 45:1140.