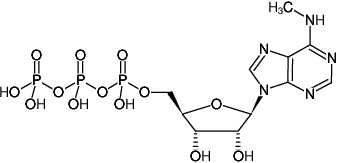
m6ATP
N6-Methyl-adenosine-5'-triphosphate, Sodium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-1101S | 10 μl (100 mM) | 55,94 | Add to Basket/Quote Add to Notepad |
| NU-1101L | 5 x 10 μl (100 mM) | 203,91 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C11H18N5O13P3 (free acid)
Molecular Weight: 521.21 g/mol (free acid)
Exact Mass: 521.01 g/mol (free acid)
CAS#: 3130-39-0 (acid)
Purity: ≥ 95 % (HPLC)
Form: solution in water
Color: colorless to slightly yellow
Concentration: 100 mM - 110 mM
pH: 7.5 ±0.5
Spectroscopic Properties: λmax 265 nm, ε 18.5 L mmol-1 cm-1 (Tris-HCl pH 7.5)
Applications:
Agonistic ligand, mainly for nucleoside receptor A1, with less affinity to A2A and A3
Nucleoside-triphosphates can be converted by different membrane-bound phosphatases into nucleosides acting as nucleoside receptor ligands. In some cases nucleoside phosphates act also directly on nucleoside receptors.
Specific Ligands:
Agonist for P2Y receptor[1] and for P2X2 purinoreceptor[2,3]
Related products: HighYield T7 RNA Synthesis Kit, #RNT-101
BIOZ Product Citations:
Selected References:
[1] Brunstock et al. (1994) Structure activity relationships for derivatives of adenosine-5'-triphosphate as agonists at P2 purinoreceptors: heterogeneity within P2X and P2Y subtypes. Drug Dev. Res. 31 (3):206.
[2] Bo et al. (1994) Comparative studies on affinities of ATP derivatives for P2X-purinoreceptors in rat urinary bladder. Br. J. Pharmacol. 112 (4):1151.
[3] He et al. (2002) Purinergic P2X (2) receptor desensitization depends on coupling between ectodomain and C-terminal domain. Molec. Pharmac. 62 (5):1187.