Kinase substrates are routinely identified by in vitro phosphorylation with radioactive ("hot") ATP whose modified gamma-phosphate (32 or 33γP) is transferred by a kinase to a serine, threonine or tyrosine hydroxyl group of its peptide/protein substrate.
Lee et al.[1] reported a non-radioactive version of in vitro phosphorylation were azide- and alkyne-γP-modified ATP analogs have been successfully used instead of 32γP-labeled ATP to phosphorylate p27kip1 with protein kinase cdk2 (Fig. 1). The phosphorylated substrate can subsequently be labeled with Biotin or fluorescent dyes via Cu(I)- catalyzed or Cu(I)-free Click-Chemistry (Tab. 1).
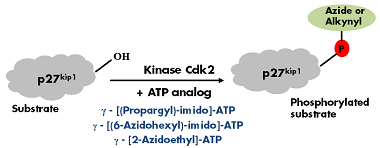
Figure 1: GST-tagged recombinant p27kip1 was incubated with kinase Cdk2 in the presence of one of the following Azide/Alkyne γP-labeled ATP analogs: γ-[(Propargyl)-imido]-ATP, γ-[(6-Azidohexyl)-imido]-ATP or γ-[2-Azidoethyl]-ATP. Phosphorylation was detected by immunoblotting with a phospho-p27kip1 specific antibody[1].
| ATP analog | Click Chemistry Type | Labeling with |
|---|---|---|
| γ-[(Propargyl)-imido]-ATP | Cu(I)-catalyzed | Azides of fluorescent dyes Azides of Biotin |
| γ-[(6-Azidohexyl)-imido]-ATP | Cu(I)-free | DBCO-containing fluorescent dyes DBCO-containing Biotin |
| γ-[2-Azidoethyl]-ATP |
[1] Lee et al. (2009) Synthesis and reactivity of novel y-phosphate modified ATP analogues. Bioorg Med Chem Lett. 19:3804.