2'/3'-O-(N-Methyl-anthraniloyl)-guanosine-5'-diphosphate, Triethylammonium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-204S | 150 μl (10 mM) | 150,00 | Add to Basket/Quote Add to Notepad |
| NU-204L | 5 x 150 μl (10 mM) | 439,20 | Add to Basket/Quote Add to Notepad |
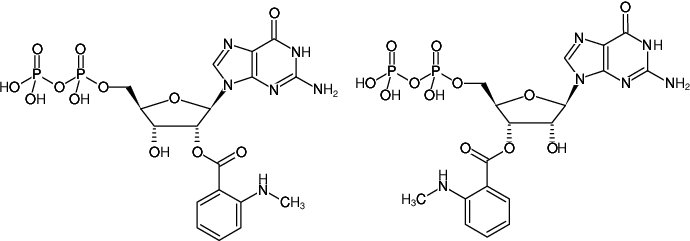
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C18H22N6O12P2 (free acid)
Molecular Weight: 576.35 g/mol (free acid)
Exact Mass: 576.08 g/mol (free acid)
CAS#: 148821-02-7
Purity: ≥ 95 % (HPLC)
Form: solution in water
Color: colorless to slightly yellow
Concentration: 10 mM - 11 mM
pH: 7.5 ±0.5
Spectroscopic Properties: λmax 252/355 nm, ε 22.6/5.7 L mmol-1 cm-1 (Tris-HCl pH 7.5), λexc 355 nm, λem 448 nm
Applications:
Conformational dynamic: DnaB/C-proteins[1], Csk[2]
Fluorescence stop-flow kinetics: release factor[3], elF5B[4], EF-1B[5], EF-Tu[6]
FRET: AC[7]
Specific Ligands:
Translation factor: elF5B[4], EF1B2[5], EF-Tu[6]
Release factor[3]
BIOZ Product Citations:
Selected References:
[1] Roychowdhury et al. (2009) Interactions of Escherichia coli DnaB-DnaC protein complex with nucleotide cofactors. 1. Allosteric conformational transitions of the complex. Biochemistry 48:6712.
[2] Shaffer et al. (2001) Nucleotide release and associated conformational changes regulate function in the COOH-terminal Src kinase, Csk. Biochemistry 40:11149.
[3] Pisareva et al. (2006) Kinetic analysis of interaction of eukariotic release factor 3 with guanine nucleotides. J. Biol. Chem. 281:40224.
[4] Pisareva et al. (2007) Kinetic analysis of the interaction of guanine nucleotides with eukariotic translation initiation factor eIF5B. Biochemistry 46:2622.
[5] Pittman et al. (2006) Mg2+ and key lysine modulate exchange activity of eukariotic translation elongation factoe 1Bα. J. Biol. Chem. 281:19457.
[6] Wieden et al. (2002) Mechanism of elongation factor (EF)-Ts-catalyzed nucleotide exchange in EF-Tu. Contribution of contacts at the guanine base. J. Biol. Chem. 277:6032.
[7] Goettle et al. (2007) Molecular analysis of the interaction of Bordetella pertussis adenylyl cyclase with fluorescent nucleotides. Molecular Pharmacology 72:526.
Wehner et al. (2012) The guanine cap of human guanylate-binding protein 1 is responsible for dimerization and self-activation of GTP hydrolysis. FEBS J. 279 (2):203.
Oreb et al. (2011) Substrate binding disrupts dimerization and induces nucleotide exchange of the chloroplast GTPase Toc33.. Biochem J. 436 (2):313.
Koshiba et al. (2011) Structure-function analysis of the yeast mitochondrial Rho GTPase, Gem1p: implications for mitochondrial inheritance. J. Biol. Chem. 286 (1):354.
Yang et al. (2010) A guaninine nucleotide exchange factor is a component of the meiotic spindle pole body in Schizosaccharomyces pombe. Mol. Biol. Cell. 21 (7):1272.
Dahl et al. (2008) The Importance of P-loop and Domain Movements in EF-Tu for Guanine Nucleotide Exchange. J. Biol. Chem. 281 (30):21139.
Ugolev et al. (2008) Dissociation of Rac1 (GDP).RhoGDI Complexes by the Cooperative Action of Anionic Liposomes Containing Phosphatidylinositol 3,4,5-Trisphosphate, Rac Guanine Nucleotide Exchange Factor, and GTP. J. Biol. Chem. 283 (32):22257.
Ugolev et al. (2008) Liposomes Comprising Anionic but Not Neutral Phospholipids Cause Dissociation of Rac (1 or 2).RhoGDI Complexes and Support Amphiphile-independent NADPH Oxidase Activation by Such Complexes. J. Biol. Chem. 281 (28):19204.
Bos et al. (2007) Structure of the cyclic-AMP-responsive exchange factor Epac2 in its auto-inhibited state. Nature 439:625.
Noel et al. (2007) Turning off the Ras switch with the flick of a finger. Nature Structural & Molecular Biology 4:677.
Pestova et al. (2006) Kinetic Analysis of Interaction of Eukaryotic Release Factor 3 with Guanine Nucleotides. J. Biol. Chem. 281 (52):40224.
Pick et al. (2006) Liposomes Comprising Anionic but not neutral Phospholipids cause dissociation of Rac (1 or 2)RhoGDI Complexes and support Amphiphile-independent NADPH oxidase Activation by Such Complexes . J. Biol. Chem. 281 (28):19204.
Wintermeyer et al. (2006) Role and timing of GTP binding and hydrolysis during EF-G-dependent RNA translocation on the ribosome. PNAS 103 (37):13670.
Thanbichler et al. (2000) Kinetics of the interaction of translation factor SelB from Escherichia coli with guanosine nucleotides and selenocysteine insertion sequence RNA. J. Biol. Chem. 275:20458.
Ahmadian et al. (1997) Individual rate constants for the interaction of Ras proteins with GTPase-activating proteins determined by fluorescence spectroscopy. Biochemistry 36:4535.
Remmers et al. (1996) Partial G protein activation by fluorescent guanine nucleotide analogs. Evidence for a triphosphatebound but inactive state. J. Biol. Chem. 271:4791.
Rensland et al. (1995) Substrate and product structural requirements for binding of nucleotides to H-ras p21: the mechanism of discrimination between guanosine and adenosine nucleotides. Biochemistry 34:593.
Richter et al. (1995) Interferon-induced MxA protein. GTP binding and GTP hydrolysis properties. J. Biol. Chem. 270:13512.
Neal et al. (1990) Hydrolysis of GTP by p21NRAS, the NRAS protooncogene product, is accompanied by a conformational change in the wild-type protein: use of a single fluorescent probe at the catalytic site. Proc. Natl. Acad. Sci. USA 87:3562.