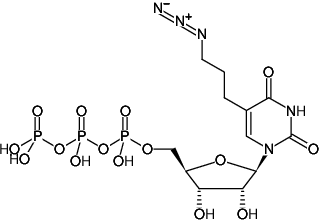
5-(3-Azidopropyl)-uridine-5'-triphosphate, Triethylammonium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-157S | 20 μl (10 mM) | 150,10 | Add to Basket/Quote Add to Notepad |
| NU-157L | 5 x 20 μl (10 mM) | 412,40 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
avoid freeze/thaw cycles
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C12H20N5O15P3 (free acid)
Molecular Weight: 567.23 g/mol (free acid)
Exact Mass: 567.02 g/mol (free acid)
Purity: ≥ 95 % (HPLC)
Form: solution in water
Color: colorless to slightly yellow
Concentration: 10 mM - 11 mM
pH: 7.5 ±0.5
Spectroscopic Properties: λ 260 nm, ε 8.8 L mmol-1 cm-1 (Tris-HCl pH 7.5)
Applications:
Incorporation into RNA by T7 RNA polymerase-mediated in vitro transcription (up to 1400 bp tested). For a comprehensive protocol please refer to PP-501 or PP-501-Cy3/Cy5.
3'-Azide-Labeling of T7 promotor-containing oligonucleotides[1,2,3]
The resulting azide-functionalized RNA can subsequently be processed via Cu(I)-free (azide-DBCO) or Cu(I)-catalyzed (azide-alkyne) click chemistry that offers the choice
- to introduce a Biotin group for subsequent purification tasks (via DBCO-functionalized Biotin or Alkynes of Biotin, respectively)
- to introduce fluorescent group for subsequent microscopic imaging (DBCO-functionalized fluorescent dyes or Alkynes of fluorescent dyes, respectively)
- to crosslink the RNA to azide- or alkyne functionalized biomolecules e.g.proteins
Related products:
BIOZ Product Citations:
Selected References:
[1] Rao et al. (2012) Enzymatic incorporation of an azide-modified UTP analog into oligoribonucleotides for post-transcriptional chemical functionalization. Nature Protocols 7 (6):1097.
[2] Rao et al. (2012) Posttranscriptional chemical functionalization of azide-modified oligoribonucleotides by bioorthogonal click and Staudinger reactions. Chem. Commun. 48 (4):498.
[3] Savant et al. (2015) A versatile toolbox for posttranscriptional chemical labeling and imaging of RNA. Nucleic Acid Res. doi: 10.1093/nar/gkv903.