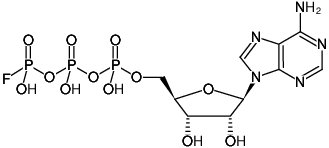
(ApppF)
Adenosine-5'-(γ-fluoro)-triphosphate, Sodium salt
Adenosine-5'-(3-fluoro)-triphosphate, Sodium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-942-5 | 5 mg | 134,30 | Add to Basket/Quote Add to Notepad |
| NU-942-25 | 25 mg | 537,40 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C10H15FN5O12P3 (free acid)
Molecular Weight: 509.17 g/mol (free acid)
Exact Mass: 508.99 g/mol (free acid)
CAS#: 37515-63-2 (free acid), 1698010-87-5 (sodium salt)
Purity: ≥ 95 % (HPLC)
Form: solid
Color: white to off-white
Spectroscopic Properties: λmax 259 nm, ε 15.4 L mmol-1 cm-1 (Tris-HCl pH 7.5)
Applications:
Substrate for snake venom phosphodiesterase [1,2]
Inhibitor of Lysyl-, Valyl- and Arginyl-tRNA synthases [3]
Substrate for Fhit proteins [1,4]
BIOZ Product Citations:
Selected References:
[1] Baranowski et al. (2015) Synthesis of fluorophosphate nucleotide analogues and their characterization as tools for ¹⁹F NMR studies. J. Org. Chem. 80 (8):3982.
[2] Haley et al. (1972) g-Fluoroadenosine triphosphate. Synthesis, properties, and interaction with myosin and heavy meromyosin. Biochemistry 11 (15):2863.
[3] Freist et al. (1980) Chemically modified ATP derivatives for the study of aminoacyl-tRNA synthetases from Bakers' yeast: ATP analogs with fixed conformations of modified triphosphate chains in the aminoacylation reaction Bioorganic Chemistry 9 (4):491.
[4] Guranowski et al. (2008) Fhit proteins can also recognize substrates other than dinucleoside polyphosphates. FEBS Lett. 582 (20):3152.