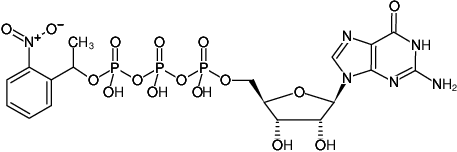
Guanosine-5'-triphosphate, P3-(1-(2-nitrophenyl)-ethyl)-ester, Sodium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NU-302S | 100 μl (10 mM) | 157,60 | Add to Basket/Quote Add to Notepad |
| NU-302L | 5 x 100 μl (10 mM) | 461,70 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
store dark
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C18H23N6O16P3 (free acid)
Molecular Weight: 672.33 g/mol (free acid)
Exact Mass: 672.04 g/mol (free acid)
CAS#: 124830-99-5
Purity: ≥ 95 % (HPLC)
Form: solution in water
Color: colorless to slightly yellow
Concentration: 10 mM - 11 mM
pH: 7.5 ±0.5
Spectroscopic Properties: λmax 254 nm, ε 16.4 L mmol-1 cm-1 (Tris-HCl pH 7.5)
Applications:
Binding to GTPase activating protein[1]
FTIR specroscopy[1, 2]
Mechanism of GTPase[2]
Enzyme kinetic studies with Ras[3]
BIOZ Product Citations:
Selected References:
[1] Allin et al. (2001) Monitoring the GAP catalyzed H-Ras GTPase reaction at atomic resolution in real time. Proc. Natl. Acad. Sci. USA 98 (14):7754.
[2] Allin et al. (2001) Ras catalyzes GTP hydrolysis by shifting negative charges from gamma- to beta-phosphate as revealed by time-resolved FTIR difference spectroscopy. Biochemistry-US 40 (10):3037.
[3] Du et al. (2001) Comparison of nitrophenylethyl and hydroxyphenacyl caging groups. Biopolymers 62:147.
Jiménez et al. (2011) Reconstitution and Organization of Escherichia coli Proto-ring Elements (FtsZ and FtsA) inside Giant Unilamellar Vesicles Obtained from Bacterial Inner Membranes . J. Biol. Chem. 286 (13): 11236
Dumas et al. (2010) Spatial regulation of membrane fusion controlled by modification of phosphoinositides. PLoS One. 5 (8):e12208.
Scheidig et al. (1999) The pre-hydrolysis state of p21 (ras) in complex with GTP: new insights into the role of water molecules in the GTP hydrolysis reaction of ras-like proteins. Struct. Fold Des. 7 (11):1311.
Gerwert (1999) Molecular reaction mechanisms of proteins monitored by time-resolved FTIR-spectroscopy. Biol. Chem. 380 (7-8):931.
Cepus et al. (1998) Time-resolved FTIR studies of the GTPase reaction of H-ras p21 reveal a key role for the beta-phosphate. Biochemistry 37 (28):10263.
Wagner et al. (1995) Interaction of guanosine nucleotides and their analogs with elongation-factor tu from thermus-thermophilus. Biochemistry 34 (39):12535.
Scheidig et al. (1995) X-ray crystal-structure analysis of the catalytic domain of the oncogene product p21 (H-ras) complexed with caged GTP and mant-dGppNHp. J. Mol. Biol. 253 (1):132.
Scheidig et al. (1994) Crystallographic studies on p21 (h-ras) using the synchrotron laue method - improvement of crystal quality and monitoring of the gtpase reaction at different time points. Acta Cryst. D 50:512.
Scheidig et al. (1992) Time-resolved crystallography on h-ras p21. Philos. T. Roy. Soc. A 340 (1657):263.
Reshetnikova et al. (1992) Crystals of intact elongation factor-Tu from thermus-thermophilus diffracting to 1.45-angstrom resolution. J. Cryst. Growth 122 (1-4):360.
Limmer et al. (1992) Nucleotide binding and gtp hydrolysis by elongation-factor Tu from thermus-thermophilus as monitored by proton NMR. Biochemistry 31 (11):2970.
Marx et al. (1990) Microtubule assembly and oscillations induced by flash-photolysis of caged-GTP. Eur. Biophys. J. 19 (1):1.
Schlichting et al. (1989) Biochemical and crystallographic characterization of a complex of c-ha-Ras p21 and caged GTP with flashphotolysis - (time-resolved structure). Proc. Natl. Acad. Sci. USA 86 (20):7687.