No login/account required.
Your Basket/Online Quote
Items: 0 (0,00 €)
» Search & Order
» Sign in / Register
Your Basket/Online Quote
Items: 0 (0,00 €)
» Search & Order
» Sign in / Register
screening kit for ATP-binding proteins
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| NK-101 | 5 ATP analogs | 461,00 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Description:
The enzymes involved in hydrolysis of nucleoside triphosphates are a major focus of biochemical research, targeting the understanding and treatment of cellular processes involved in diseases such as cancer, viral infections, cardiovascular and motional disorders. To elucidate the mechanisms of action of these enzymes, it is often desirable to trap the enzymes in their triphosphate-bound state. Since natural nucleotides are hydrolyzed at rates usually preventing such studies, nucleotide analogs are used that are hydrolyzed at much smaller rates or that are even completely resistant to hydrolysis.
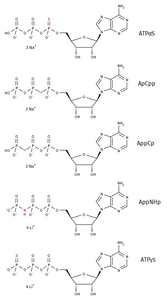
The Non-hydrolyzable ATP Test Kit contains a set of 5 typical non-hydrolyzable Adenosine-5'-triphosphate analogs.
The position of the modification within the triphosphate exerts influence on its binding affinity to a particular enzyme and its hydrolysis rate.
This Kit is designed to identify the most suitable non-hydrolyzable ATP derivative for your application.
Content:
| Non-hydrolyzable Adenosine Nucleotide | Cat. No. | Amount |
| ATPαS | NU-408S | 25 μl (100 mM) |
| ApCpp* | NU-421-2 | 2 mg |
| AppCp* | NU-422-10 | 10 mg |
| AppNHp* | NU-407-10 | 10 mg |
| ATPγS* | NU-406-5 | 5 mg |
BIOZ Product Citations:
Selected References:
Toyoshima et al. (2004) Crystal structure of the calcium pump with a bound ATP analogue. Nature 430:529.
Bivalacqua et al. (2002) Comparative responses to alpha,beta-methylene-ATP in cat pulmonary, mesenteric, and hindquarter vascular beds. Appl. Physiol. 93 (4):1287.
Yengo et al. (2001) Myosin V is weakly bound to actin in the presence of ATPgammas. Biophys. J. 80 (1):2609, Part 2.
Shi et al. (1999) EDC cross-linking of actin and myosin S1. II. AMPPNP induces an allosteric transition from pre-hydrolysis state to posthydrolysis state. Biophys. J. 76 (1):A163.
OsumiDavis et al. (1996) Effect of alpha-Thio-ATP on the steady-state kinetics of nucleotide incorporation catalyzed by the wildtype and active-site mutant T7 RNA polymerases. Biophys. J. 70 (2):SU480, Part 2.