Di-adenosine polyphosphates (ApnAs) add to a fast growing list of nucleotide derivatives (such as c-di-GMP or c-di-AMP) with prospective signaling functions in bacteria[1]. Originating as side products of tRNA aminoacylation (Scheme 1), ApnAs have been implicated in the regulation of stress response[2], biofilm formation[3], pathogenesis[4] and antibiotic resistance[5]. Current research is aiming to establish the elusive mechanistic aspects of ApnA action. Recently, Jena Biosciences ApnAs have found their application as
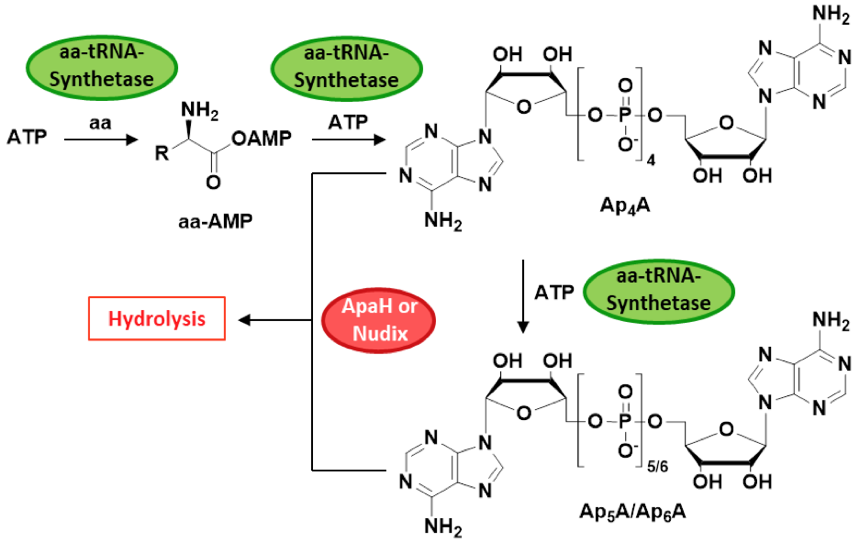
Scheme 1: Biosynthesis and clearance of ApnAs. Di-adenosine polyphosphates are by-products of the tRNA-aminoacylation pathway. In the presence of ATP, amino acids (aa) are adenylated by the corresponding aminoacyl-tRNA synthetase. Transfer of the activated AMP moiety onto ATP yields Ap4A, which serves as a substrate for further phosphate transfer cycles, thereby generating Ap5A and Ap6A. ApnA-cleavage is catalyzed by ApaH or Nudix hydrolases.
| Compound | Cat. No. | Amount | Price (€) |
|---|---|---|---|
| AP3A - Solution | NU-506S | 50 μl (10 mM) | 112,80 |
| NU-506L | 5x 50 μl (10 mM) | 330,42 | |
| AP3A - Solid | NU-506-5 | 5 mg | 101,00 |
| NU-506-25 | 25 mg | 404,00 | |
| AP2A | NU-936-1 | 5 mg | 101,00 |
| NU-936-5 | 25 mg | 404,00 | |
| AP4A - Solution | NU-507S | 100 μl (10 mM) | 112,80 |
| NU-507L | 5x 100 μl (10 mM) | 330,42 | |
| AP4A - Solid | NU-507-5 | 5 mg | 101,00 |
| NU-507-25 | 25 mg | 404,00 | |
| AP5A - Solution | NU-508S | 50 μl (10 mM) | 118,45 |
| NU-508L | 5x 50 μl (10 mM) | 346,95 | |
| AP6A - Solution | NU-509S | 20 μl (10 mM) | 112,80 |
| NU-509L | 5x 20 μl (10 mM) | 330,42 |
Non-hydrolyzable ApnA derivatives can be obtained as custom synthesis products. Please inquire!
[1] Kisselev et al. (1998) Diadenosine oligophosphates (Ap(n)A), a novel class of signalling molecules? FEBS Lett. 427 (2):157.
[2] Bochner et al. (1984) AppppA and related adenylylated nucleotides are synthesized as a consequence of oxidation stress. Cell 37:225.
[3] Monds et al. (2010) Di-adenosine tetraphosphate (Ap4A) metabolism impacts biofilm formation by Pseudomonas fluorescens via modulation of c-di-GMP-dependent pathways. J. Bacteriol. 192:3011.
[4] Ismail et al. (2003) Regulation of dinucleoside polyphosphate pools by the YgdP and ApaH hydrolases is essential for the ability of Salmonella enterica serovar Typhimurium to invade cultured mammalian cells. J. Biol. Chem. 278:32602.
[5] Hansen et al. (2008) Role of global regulators and nucleotide metabolism in antibiotic tolerance in Escherichia coli. Antimicrob. Agents Chemother. 52:2718.
[6] Kimura et al. (2017) High concentrations of intracellular Ap4A and/or Ap5A in developing Myxococcusxanthus cells inhibit sporulation. Microbiology 163:86.
[7] Nguyen et al. (2016) Substrate specificity characterization for eight putative nudix hydrolases. Evaluation of criteria for substrate identification within the Nudix family. Proteins 84 (12):1810.
[8] Oka et al. (2016) Lysyl-tRNA synthetase from Myxococcus xanthus catalyzes the formation of diadenosine penta- and hexaphosphates from adenosine tetraphosphate. Arch. Biochem. Biophys. 604:152
[9] Anashkin et al. (2015) Cystathionine β-Synthase (CBS) Domain-containing Pyrophosphatase as a Target for Diadenosine Polyphosphates in Bacteria. J. Biol. Chem. 290 (46):27594.
[10] Despotović et al. (2017) Diadenosine tetraphosphate (Ap4A) - an E. coli alarmone or a damage metabolite? FEBS J. 284 (14):2194.