Highly specific peptidases are especially important in the field of Digestions Proteomics to guarantee well defined peptide fragments. One of such peptidases is human Legumain, a strictly specific enzyme for the hydrolysis of peptide bonds after asparagines and, to a lesser extent, aspartates in a pH dependent manner (figure A). It is therefore also known as the asparaginyl endopeptidase (AEP).
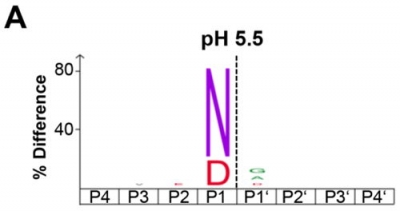
Figure A: iceLogo showing that Legumain specifically cleaves substrates after asparagine and, to a lesser extent, aspartic acid residues at pH 5.5.
In human, Legumain is synthesized as an inactive proenzyme consisting of a catalytic domain and a LSAM prodomain (Legumain Stabilization and Activity Modulation domain), linked by an activation peptide (AP). For activation, the prodomain needs to be autocatalytically cleaved by lowering the pH (figure B). While Prolegumain is stable at neutral pH, isolated active Legumain is only stable at acidic conditions. We offer ready-to-use Legumain, already cleaved and stored under buffer conditions, guaranteeing full activity.
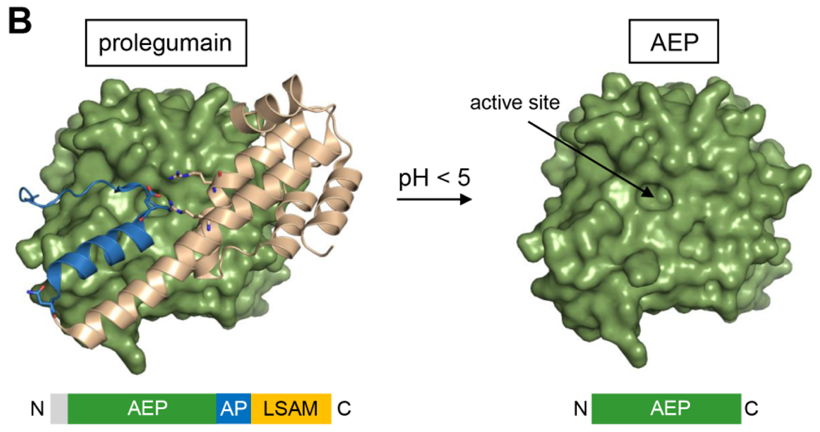
Figure B: Prolegumain is activated by autocatalytic removal of the C-terminal prodomain at acidic pH.
Legumain is also interesting on a pathophysiological level as it is overexpressed in the majority of human solid tumors, including breast and colorectal carcinoma, making it a perfect candidate for targeted drug delivery approaches. Normally Legumain is involved in initiating immune response in the endosomal system. In tumors however, it is associated with enhanced tissue invasion and metastasis activity and its overexpression correlates with poor outcome prognosis.
Due to its strict substrate specificity and pathophysiological relevance, Legumain attracts growing interest as an enzyme for digestion proteomics and targeted drug delivery. Jena Bioscience offers activated human Legumain, resulting from recombinant expression of Prolegumain in Leishmania and consequent activation under acidic conditions.
| Product | Cat. No. | Amount | Price |
|---|---|---|---|
| Legumain Asparaginyl Endopeptidase (AEP) human, recombinant, Leishmania |
PR-967S | 10 µg | 293,40 € |
| PR-967L | 5 x 10 µg | 1.173,40 € | |
| PR-967XL | no longer available |
Dall et al. (2013) Mechanistic and structural studies on legumain explain its zymogenicity, distinct activation pathways, and regulation. Proceedings of the National Academy of Sciences of the United States of America 110 (27):10940.
Dall et al. (2015) Structure and mechanism of an aspartimide-dependent peptide ligase in human legumain. Angew Chem Int Ed Engl 54 (10):2917.
Soh et al. (2020) ExteNDing Proteome Coverage with Legumain as a Highly Specific Digestion Protease. Anal Chem 92 (4):2961.