Integral membrane proteins are hard to express with common recombinant expression systems. Learn how LEXSY can help!
The eukaryotic expression system LEXSY was used to produce Channelrhodopsin 2 (ChR2) both in its native form and as a slow-mutant with yields in the mg per litre range. The recombinant proteins were crystallized and their crystal structures solved at 2.4 and 2.7 Å, respectively (Figure 1)1.
While membrane protein expression is one bottleneck, crystallization is another and usually requires either detergents for solubilized membrane proteins or lipids to create artificial membranes such as the lipidic cubic phase (LCP) that can be found in our section Screening Membrane Proteins.
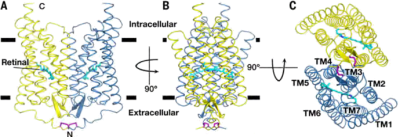
Figure 1: Overall structure presentation of the channelrhodopsin ChR2 dimer. A to C = view from different angles; TM1 to TM7 = transmembrane helices; Retinal = chromophore; cysteine bridges are shown in purple. Adapted from Volkov et al., 2017.
LEXSY Expression Kits
Convenient all-in-one expression kits allowing constitutive or inducible, intracellular or secretory expression of target proteins. This is the place to start if you are new to LEXSY.
LEXSY Strains & Cultivation Kits
The LEXSY expression hosts, as strains or complete ready-to-grow cultivation kits.
LEXSY Cultivation Media
Complex and synthetic media for optimal growth of LEXSY strains.
LEXSY Antibiotics
For efficient selection of recombinant strains. Also useful for more than 100 other cell lines.
LEXSY Expression Vectors
Available with six different selection markers.
PCR and Cloning enzymes
All restriction enzymes for the pLEXSY cloning sites, ligases, polymerases etc.

Need further information? Please check out the LEXSY section or contact Andreas by email at expression@jenabioscience.com !
Volkov et al. (2017) Structural insights into ion conduction by channelrhodopsin 2. Science 358:1018.