Post-translational modifications (PTMs) like glycosylation are often essential for the correct folding and function of proteins. Prokaryotic expression systems like E. coli cannot perform these modifications, which can lead to inactive or misfolded proteins and expression in inclusion bodies, requiring laborious refolding.
The eukaryotic expression system LEXSY is able to carry out many post-translational protein modifications while retaining the ease of handling of prokaryotic systems. The most notable PTM is a human-like glycosylation pattern, but more than 10 other modifications have been achieved with LEXSY.
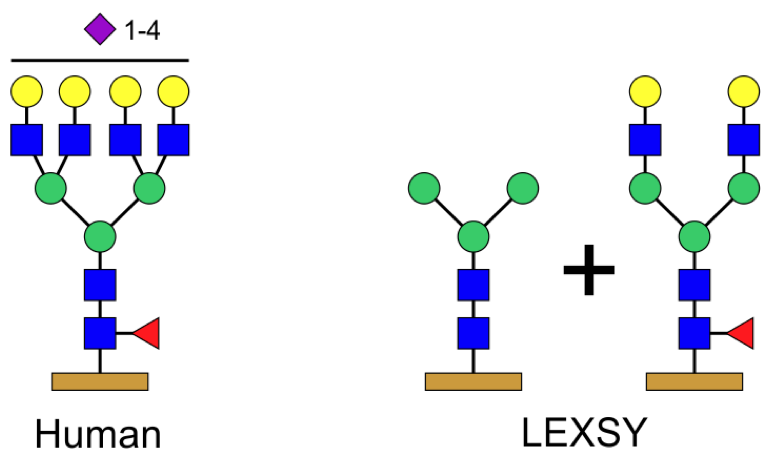
Figure 1: Comparison between the glycosylation patterns of human and LEXSY-produced proteins.
| Modification | Selected references |
|---|---|
| Disulfide bond formation | Hong et al. 2008[1] |
| Multimerization | Phan et al. 2009[2] |
| Proteolytic processing | Basak et al. 2008[3] |
| Glycosylation | Breitling et al. 2002[4] |
| Glypiation | Basak et al. 2008[3] |
| Prenylation | Gillespie et al. 2007[5] |
| Methylation | Rosenzweig et al. 2008[6] |
| Phosphorylation | Rosenzweig et al. 2008[6] |
| Acetylation | Gazdag et al. 2010[7] |
| Myristoylation | Sahin et al. 2008[8] |
| Amidation | Liu et al. 2000[9] |
| Glutathionylation | Melchers et al. 2007[10] |
| ADP-Ribosylation | Sahin et al. 2008[8] |
Need further information? Please check out the LEXSY section or contact us!
[1] Hong et al. (2008) Identification and enzymatic activities of four protein disulfide isomerase (PDI) isoforms of Leishmania amazonensis. Parasitol. Res. 102:437.
[2] Phan et al. (2009) The production of recombinant human laminin-332 in a Leishmania tarentolae expression system. Protein Expr. Purif. 68:79.
[3] Basak et al. (2008) Recombinant proprotein convertase 4 (PC4) from Leishmania tarentolae expression system: Purification, biochemical study and inhibitor design. Protein Expr. Purif. 60:117.
[4] Breitling et al. (2002) Non-pathogenic trypanosomatid protozoa as a platform for protein research and production. Protein Expr. Purif. 25:209.
[5] Gillespie et al. (2007) C-terminal proteolysis of prenylated proteins in trypanosomatids and RNA interference of enzymes required for the post-translational processing pathway of farnesylated proteins. Mol. Biochem. Parasitol. 153:115.
[6] Rosenzweig et al. (2008) Post-translational modification of cellular proteins duringLeishmania donovani differentiation. Proteomics 8:1843.
[7] Gazdag et al. (2010) Purification and crystallization of human Cu/Zn superoxide dismutase recombinantly produced in the protozoan Leishmania tarentolae. Acta Crystallogr. Sect. F. Struct. Biol. Cryst. Commun. 66:871.
[8] Sahin et al. (2008) The Leishmania ARL-1 and Golgi Traffic. PLoS One 3:e1620.
[9] Liu et al. (2000) Isolation and analysis of a new developmentally regulated gene from amastigotes of Leishmania mexicana mexicana. Parasitol. Res. 86:140.
[10] Melchers et al. (2007) Glutathionylation of Trypanosomal Thiol Redox Proteins. J. Biol. Chem. 282:8678.