| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| CO-501 | 24 glass beads (each in a 1,5 ml tube) | 64,90 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped at ambient temperature
Storage Conditions: store at ambient temperature
Shelf Life: n/a
Applications:
Preparation of seed stocks from protein crystals for microseeding applications. A highly polished glass bead and a microcentrifuge tube are used as mortar and pestle for crushing of seed crystals.
Content:
24 glass beads, each in a 1.5 ml microcentrifuge tube.
Background
Seeding experiments allow to improve the likelihood of successful protein crystallization and to optimize the growth conditions.
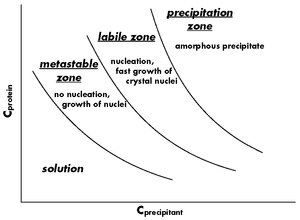
In order to crystallize a biological macromolecule, its concentration is slowly increased until a point of supersaturation is reached. Using the phase diagram,
supersaturation can be displayed in three successive zones, i.e. metastable supersaturation, labile supersaturation and precipitation (Fig. 1). In the metastable zone, no spontaneous nucleation can occur but crystals added to this zone can grow. In the labile zone, spontaneous nucleation takes place and fast growth of the nuclei is observed. In the precipitation zone, the biological macromolecule is many times supersaturated leading to a formation of amorphous precipitate.
Nucleation occurs at a higher level of supersaturation than crystal growth. By placing seeds into a solution supersaturated in the metastable zone, the growth conditions can be optimized and large single crystals can be obtained.
The number of crystals grown can be influenced by the concentration of the seed stock which is added to the protein drop. Seeding with a very concentrated seed stock can result in showering of microcrystals. If the seed stock is too dilute, no nuclei will be transferred to the protein drop. The ideal concentration of the seeding solution can be determined experimentally by performing serial dilutions from a concentrated seed stock.
Instructions for preparation of a seed stock
Please note: If your stabilization solution contains detergents or other additives which may foam vortexing is not recommended. Alternatively, the sample is treated exactly as described above, but instead of vortexing it is placed in an ultrasonic cleaner for two 1 min intervals.
Use a new microcentrifuge tube and a clean glass bead each time you prepare a new seed stock to avoid contamination and carryover from past experiments.
Performing the crystallization experiment
Prepare the crystallization drop by mixing protein sample and seed stock. Do not add reservoir solution since this may dissolve the seeds. Place the drops over a reservoir solution which has the same composition as the reservoir that was used to grow the initial seed crystal.
Ideally, the seeds should be transferred to a protein solution in the metastable zone, i.e. the solution should be slightly supersaturated.
Transferring seeds to an under saturated solution would result in the dissolution of the nuclei and no crystal growth would be observed. Transferring seeds to a highly supersaturated solution can yield showers of microcrystals.
BIOZ Product Citations:
Selected References:
Luft et al. (1999) A method to produce microseed stock for use in the crystallization of biological macromolecules. Acta Cryst. D 55:988.