Current near-IR fluorescent RNA probes use dye-labeled nucleotides with only moderate sensitivity.
Use copper-free CLICK labeling for increased sensitivity. Learn more below!
Near-infrared (IR) fluorescent RNA probes are valuable tools for non-radioactive Northern Blot experiments due to a low auto-fluorescence background and thus a high signal-to-noise-ratio within the near-infrared spectrum (650 – 900 nm).
Production of near-infrared (IR) fluorescent RNA probes is currently based on enzymatic incorporation of correspondingly labeled nucleotides (e.g. ATTO680-labeled UTP) that generate labeled probes with only moderate sensitivity (~ 1 fmol, Köhn et al. (2010)) compared to traditional 32P-labeling (< 0.01 fmol).
Miller et al. (2018) reported a non-radioactive alternative for the production of near-IR dye-labeled RNA probes with a detection limit of up to 0.05 fmol (Fig. 1).
Azide-functionalized RNA probe are efficiently produced by in vitro transcription (HighYield T7 RNA Azide Labeling Kit) and subsequently labeled with DBCO-functionalized IRDye® via copper-free CLICK chemistry (Fig. 1A). A high labeling efficiency and thus increased sensitivity is achieved due to the excellent enzymatic substrate properties of Azide-functionalized UTP (Fig. 1C).
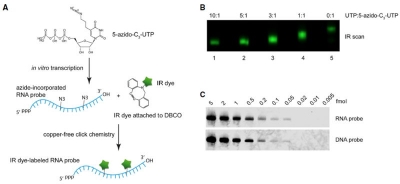
Figure 1: Near-IR dye-labeled RNA probes with a detection sensitivity of up to 0.05 fmol are produced via in vitro transcription followed by copper-free CLICK labeling (according to Miller et al. (2018)).
(A) Principle of near-IR dye-labeled RNA probe production. (B) Analysis of optimal UTP:5-Azido-C3-UTP ratio during in vitro transcription (IR scan). C) Northern Blot analysis of Ta U6 snRNA (serial dilution) with IRDye® 800C-labeled RNA probe.
HighYield in vitro transcription kits for preparation of Digoxigenin- and Biotin- labeled RNA probes are available as well!

Please contact Barbara with all questions or inquiries you may have!