No login/account required.
Your Basket/Online Quote
Items: 0 (0,00 €)
» Search & Order
» Sign in / Register
Your Basket/Online Quote
Items: 0 (0,00 €)
» Search & Order
» Sign in / Register
Regulation of the cellular machinery requires both cleavage and transfer of phosphate bonds by ATPases, G-proteins, kinases, and polymerases. By replacing the 5'-triphosphate moieties of ATP and GTP for electrophilic 4-fluorosulfonylbenzoyl (FSB) residues, the corresponding analogs FSBA[1] and FSBG[2] have been devised as
Furthermore, recent advances in the field of SuFEx-Chemistry[7] allow us to access numerous sulfonyl fluoride motifs. Please inquire for structures.
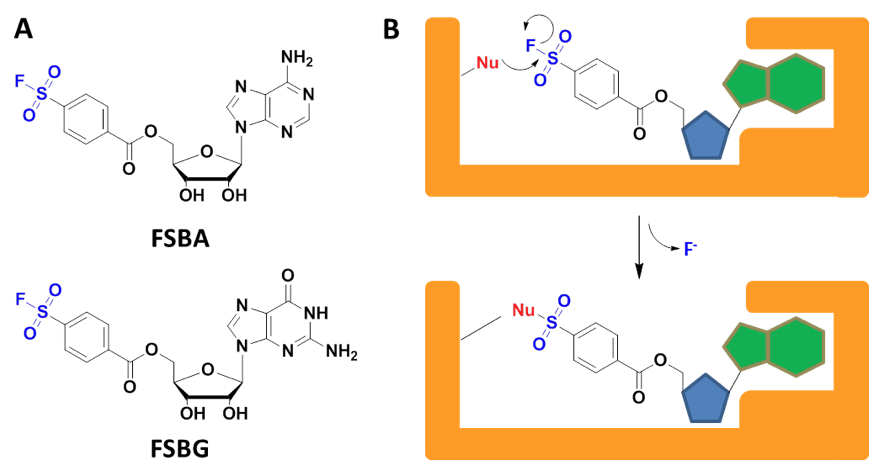
Scheme 1: A) The structures of FSBA and FSBG with the reactive sulfonyl fluoride moieties highlighted in blue. B) Sulfonyl fluorides are molecular warheads[8] with sufficient stability in aqueous solution under physiological conditions. When exposed to an activated nucleophile such as serine, threonine, lysine, tyrosine or histidine at the active site of a target enzyme, covalent adducts are formed, permitting the context-specific probing of functionally important residues.
[1] Pal et al. (1975) Affinity labeling of the inhibitory DPNH site of bovine liver glutamate dehydrogenase by 5'-fluorosulfonylbenzoyl adenosine. J. Biol. Chem. 250 (20):8140.
[2] Pal et al. (1978) A new affinity label for guanosine nucleotide sites in proteins. J. Biol. Chem. 253 (19):6644.
[3] Wyatt et al. (1977) Affinity labeling of rabbit muscle pyruvate kinase by 5'-p-fluorosulfonylbenzoyladenosine. Biochemistry 16 (7):1333.
[4] Bretner et al. (2004) Synthesis and evaluation of ATP-binding site directed potential inhibitors of nucleoside triphosphatases/helicases and polymerases of hepatitis C and other selected Flaviviridae viruses. Antivir. Chem. Chemother. 15 (1):35.
[5] Moore et al. (2004) Anti-sulfonylbenzoate antibodies as a tool for the detection of nucleotide-binding proteins for functional proteomics. J. Proteome. Res. 3 (6):1184.
[6] Embogama et al. (2017) K-BILDS: A Kinase Substrate Discovery Tool. ChemBiochem. 18 (1):136.
[7] Dong et al. (2014) Sulfur(VI) fluoride exchange (SuFEx): another good reaction for click chemistry. Angew. Chem. Int. Ed. Engl. 53 (36):9430.
[8] Narayanan et al. (2015) Sulfonyl fluorides as privileged warheads in chemical biology. Chem. Sci. 6:2650.