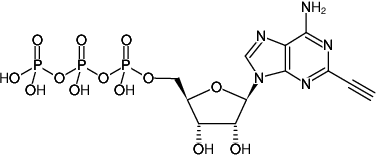
2-Ethynyl-adenosine-5’-triphosphate, Sodium salt
| Cat. No. | Amount | Price (EUR) | Buy / Note |
|---|---|---|---|
| CLK-NU-004S | 100 μl (10 mM) | 150,00 | Add to Basket/Quote Add to Notepad |
| CLK-NU-004L | 5 x 100 μl (10 mM) | 439,00 | Add to Basket/Quote Add to Notepad |
For general laboratory use.
Shipping: shipped on gel packs
Storage Conditions: store at -20 °C
Short term exposure (up to 1 week cumulative) to ambient temperature possible.
Shelf Life: 12 months after date of delivery
Molecular Formula: C12H16N5O13P3
Molecular Weight: 531.20 g/mol
Exact Mass: 531.00 g/mol
Purity: ≥ 90 % (HPLC), contains approx. 6 % 2-Ethynyl-ADP
Form: solution in 100 mM Tris-HCl
Color: colorless to slightly yellow
Concentration: 10 mM - 11 mM
pH: 7.5 ±0.5
Spectroscopic Properties: λmax 265 nm, ε 10.6 L mmol-1 cm-1 (Tris-HCl pH 7.5)
Applications:
in vitro polyadenylation of RNA[1]
Description:
2-Ethynyl-labeled adenosine triphosphate (2-EATP) is suitable for in vitro polyadenylation of RNA with recombinant poly(A) polymerase[1].
The resulting Alkyne-functionalized RNA can subsequently be processed via Cu(I)-catalyzed Azide-Alkyne click
chemistry (CUAAC) that offers the choice
Presolski et al.[2] and Hong et al.[3] provide a general protocol for Cu(I)-catalyzed click chemistry reactions that may be used as a starting point for the set up and optimization of individual assays.
Related products:
BIOZ Product Citations:
Selected References:
[1] Curanovic et al. (2013) Global profiling of stimulus-induced polyadenylation in cells using a poly (A) trap. Nat. Chem. Biol. 9:671.
[2] Presolski et al. (2011) Copper-Catalyzed Azide-Alkyne Click Chemistry for Bioconjugation. Current Protocols in Chemical Biology 3:153.
[3] Hong et al. (2011) Analysis and Optimization of Copper-Catalyzed Azide-Alkyne Cycloaddition for Bioconjugation. Angew. Chem. Int. Ed. 48:9879.