Research on bacterial second messengers has recently experienced tremendous progress, and cyclic di-AMP (c-di-AMP) is one of the rising stars.[1] c-di-AMP is essential for the growth of several human bacterial pathogens - e.g. Staphylococcus aureus or Borrelia burgdorferi - by regulating DNA damage repair, potassium homeostasis, cell wall and membrane synthesis and sporulation.[2] To solve the numerous riddles relating to c-di-AMP metabolism and function (Scheme 1)[2], synthetic c-di-AMP serves as:
c-di-AMP is now available from Jena Bioscience. For other cyclic or linear dinucleotide pairs, please inquire!
| Compound | Cat. No. | Amount | Price (€) |
|---|---|---|---|
| cyclic-di-AMP (c-di-AMP) | NU-954S | 1 μmol | 95,00 |
| NU-954L | 5 x 1 μmol | 380,00 |
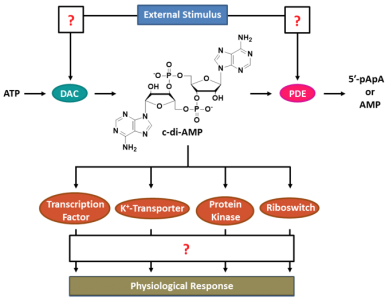
Scheme 1: The c-di-AMP signaling network. Biosynthesis and degradation of c-di-AMP are triggered by external stimuli that modulate the activities of diadenylate cyclases (DACs) and phosphodiesterases (PDEs) in a widely elusive manner. DACs catalyze the cyclization of two ATP molecules, yielding c-di-AMP that acts as a ligand of transcription factors, potassium transporters, protein kinases and RNA riboswitches. However, most regulatory effects of c-di-AMP-binding are poorly understood and subject to current investigations. In the PDE-catalyzed clearance reaction, c-di-AMP is hydrolyzed to 5'-pApA or AMP.
Selected References:
[1] Corrigan et al. (2013) Cyclic di-AMP: another second messenger enters the fray. Nat. Rev. Microbiol. 11 (8):513.
[2] Fahmi et al. (2017) c-di-AMP: An Essential Molecule in the Signaling Pathways that Regulate the Viability and Virulence of Gram-Positive Bacteria. Genes 8 (8):197.
[3] Bowman et al. (2016) New Insights into the Cyclic Di-adenosine Monophosphate (c-di-AMP) Degradation Pathway and the Requirement of the Cyclic Dinucleotide for Acid Stress Resistance in Staphylococcus aureus. J. Biol. Chem. 291 (53):26970.
[4] He et al. (2016) Structural and Biochemical Insight into the Mechanism of Rv2837c from Mycobacterium tuberculosis as a c-di-NMP Phosphodiesterase. J. Biol. Chem. 291 (7):3668.
[5] Sureka et al. (2014) The cyclic dinucleotide c-di-AMP is an allosteric regulator of metabolic enzyme function. Cell 158 (6):1389.
[6] Wang et al. (2018) Structural and biochemical characterization of the catalytic domains of GdpP reveals a unified hydrolysis mechanism for the DHH/DHHA1 phosphodiesterase. Biochem. J. 475 (1):191.
[7] Schuster et al. (2016) The second messenger c-di-AMP inhibits the osmolyte uptake system OpuC in Staphylococcus aureus. Sci. Signal. 9 (441):ra81.
[8] Drexler et al. (2017) Structural and Biophysical Analysis of the Soluble DHH/DHHA1-Type Phosphodiesterase TM1595 from Thermotoga maritima. Structure 25 (12):1887.
[9] Xia et al. (2018) The ER membrane adaptor ERAdP senses the bacterial second messenger c-di-AMP and initiates anti-bacterial immunity. Nat. Immunol. 19 (2):141.